 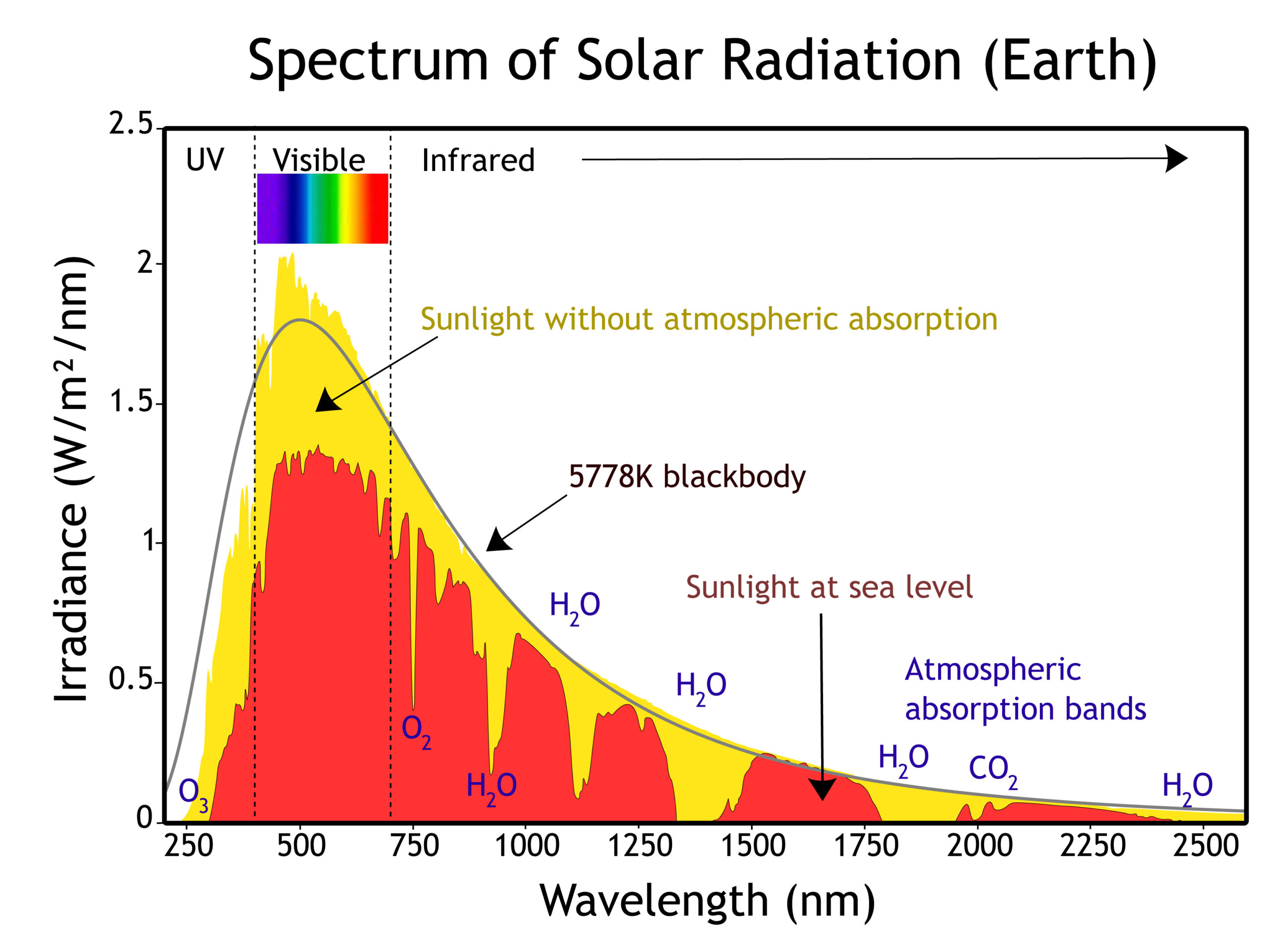
Therefore, when this light passes through a gas, the gas atoms may absorb certain wavelengths to produce a line absorption spectrum.īlack hydrogen absorption lines are at the same wavelength as the bright hydrogen emission lines. This is called a line absorption spectrum. If anybody can point me in the right direction it would be greatly appreciated. So if a continuous spectrum travels through a cooler gas, it will ‘pull out’ or absorb certain wavelengths of light to produce a series of dark lines on a continuous spectrum of light. 1 sheenarbw 7 0 I've been looking everywhere for information on the absorption spectra of molecular hydrogen. When a gas is cool, it absorbs the same wavelengths of light as it would emit when it is hot. A gas cloud on its own, without a light source behind it, produces a line emission spectrum. Hot gases don’t produce a continuous emission spectrum.Ī hot gas only emits certain wavelengths of light to produce bright lines on a dark background. When a gas is very hot, it doesn’t emit all wavelengths of light. A light source, such as a star or a filament bulb, gives a continuous emission spectrum. This is called a continuous emission spectrum. The concentration information is often retrieved by fitting absorption profiles to the acquired spectra, utilizing spectroscopic databases. This light, when seen through a prism or diffraction grating, shows all wavelengths of visible light. Infrared absorption spectroscopy is a widely used tool to quantify and monitor compositions of gases. Focusing effects associated with these bifurcations cause some recurrences to be particularly strong.Studying the line spectra produced by hot gases and absorbed by cooler gases allows us to identify the elements in stars. This continuous absorption corresponds to the dissociation of the molecule into a normal and an excited. These bifurcations can have generic structure, or sometimes the structures are modified by symmetries of the system. A continuous absorption spectrum begins sharply at 840. Other recurrences are created by period-tripling and higher-order bifurcations of existing orbits. The "main sequence" of orbits is produced from an orbit parallel to B through a sequence of pitchfork and period-doubling bifurcations. New "exotic" orbits suddenly appear "out of nowhere" through saddle-node bifurcations. Bifurcation theory provides organizing principles for understanding this proliferation and for interpreting the data. as the scaled energy is increased, observed recurrences proliferate, consistent with the change from orderly to chaotic motion of the electron. Closed-orbit theory gives formulas for these recurrence strengths. There are more lines in the absorption spectrum at low temperature (Raising the temperature of the hydrogen. However, the processes by which these two spectra are generated are completely the opposite of each other. What is the result of such a comparison Answer: C. So, naturally, if an atom exhibits some emission spectrum, it will also give an absorption spectrum. A Fourier transformation of this signal yields peaks which we interpret as "recurrence strengths" which depend upon the classical action of the closed orbit. Emission and absorption are inverse processes. (Recall from the Formation of Spectral Lines section, the lines are the result of electrons in orbit around a nucleus changing energy levels. The absorption rate was observed to exhibit sinusoidal fluctuations which we correlate with closed classical orbit of the electron. Because the electron and the proton are separated, ionized hydrogen cannot produce absorption lines. An absorption spectrum results when light from a continuous source passes through a cooler gas, consisting of a series of dark lines characteristic of the composition of the gas. Taking advantage of a classical scaling law, the photon energy and the magnetic field strength were varied simultaneously in the experiment and the absorption rate vs. An emission spectrum can be produced by a gas at low pressure excited by heat or by collisions with electrons. The measurements, performed by the Bielefeld, Germany experimental group, investigated the photoabsorption to levels near the ionization threshold in magnetic fields ranging from 2.7 to 6 Tesla. Measurements of the absorption spectrum of atomic H in strong magnetic fields have been analyzed. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
